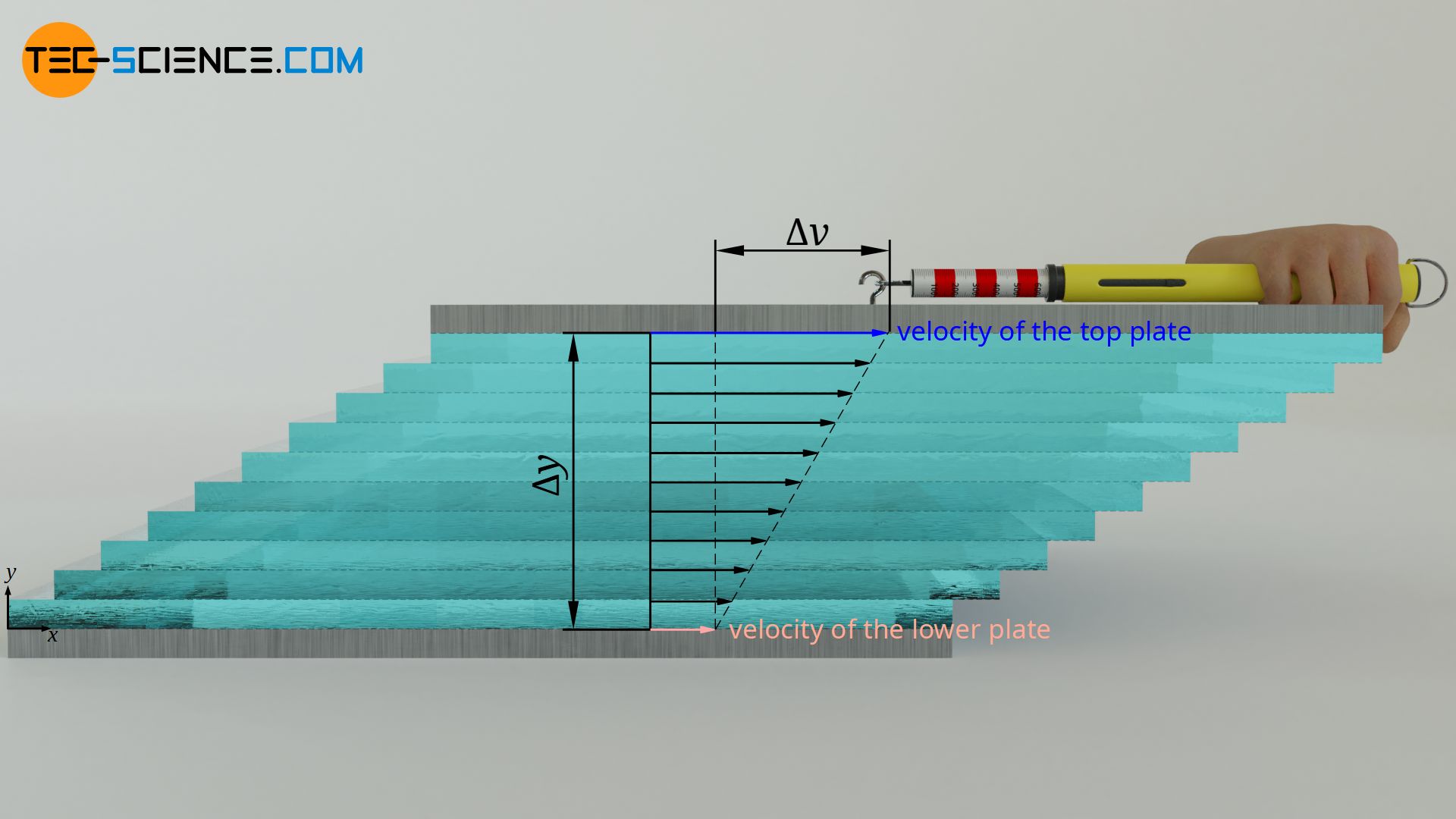
 Surface tension = Force per unit length in N/m. Surface tension has the unit of force/unit length or energy/unit area. This liquid property is known as surface tension. Adhesion allows it to adhere to another body.Īt the interface between a liquid and a gas, that is, in a liquid, the out-of-equilibrium force between molecules creates an imaginary surface layer, which exerts a tension force on the surface.Cohesion allows a liquid to resist tensile stress.Liquids have both cohesion and adhesion and both are forms of molecular attraction. It is also described as the tensile force on the surface of the liquid in contact with a gas, such that the contact surface acts as a membrane under tension. The surface energy per unit area of the interface is known as the surface tension coefficient of surface tension. Read Also: What are the Types of Fasteners? Their Uses & Examples #6 Surface TensionĪ liquid creates an interface with the second liquid or gas. Specific weight or weight density of the fluid.The following are the different properties of fluids: You might like: What are Flow Control Valves? Properties of FluidsĪn understanding of these properties is necessary for us to implement the basic principle of fluid mechanics in the solution of practical problems. Fluid dynamics includes well-known subfields like aerodynamics and hydrodynamics. Fluids dynamics: It includes the study of fluid motion and flow. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
